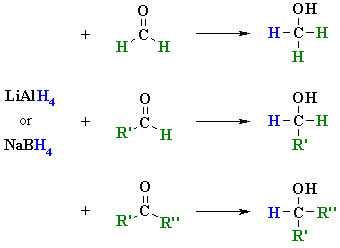First Class Tips About How To Reduce Carbonyl

Its expiry date is 2003 but the bottle is almost unused.
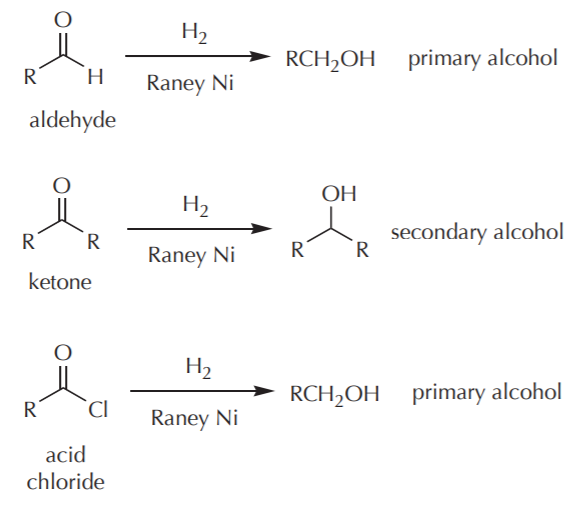
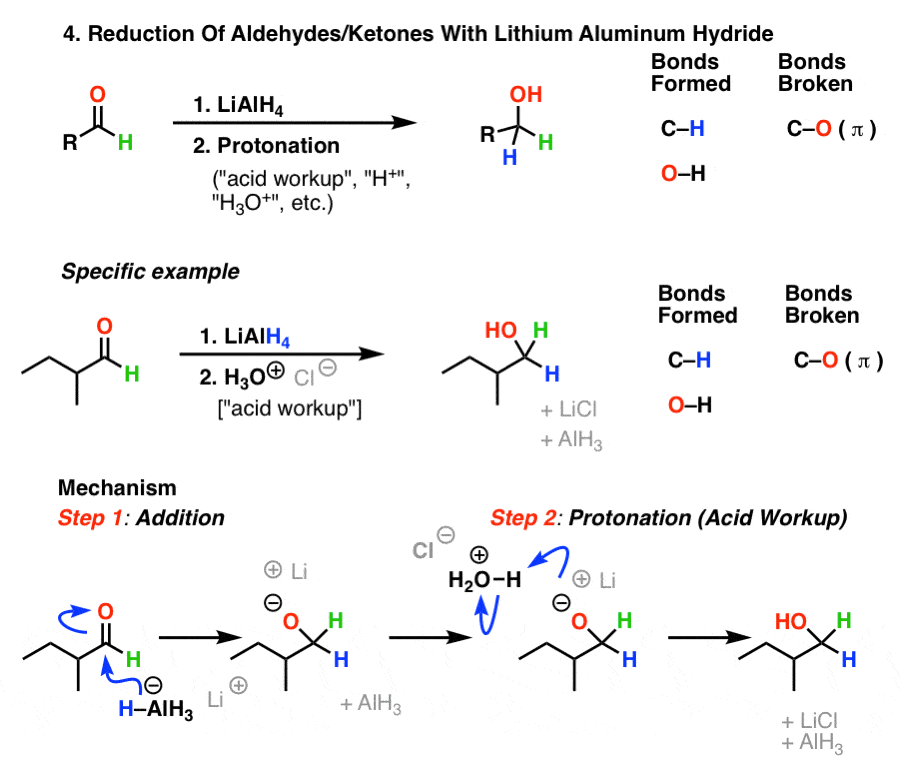
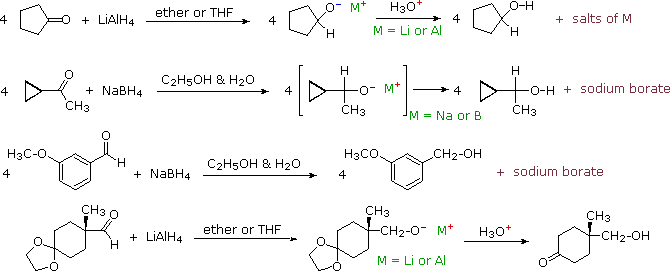
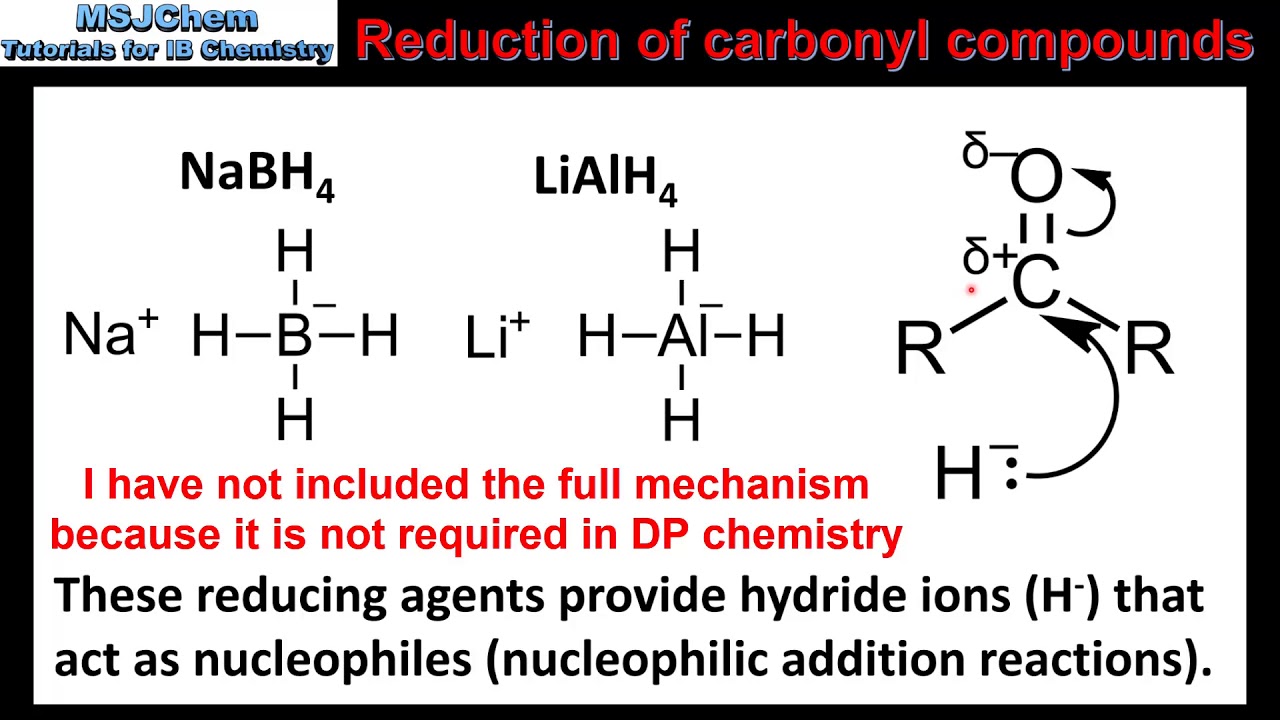
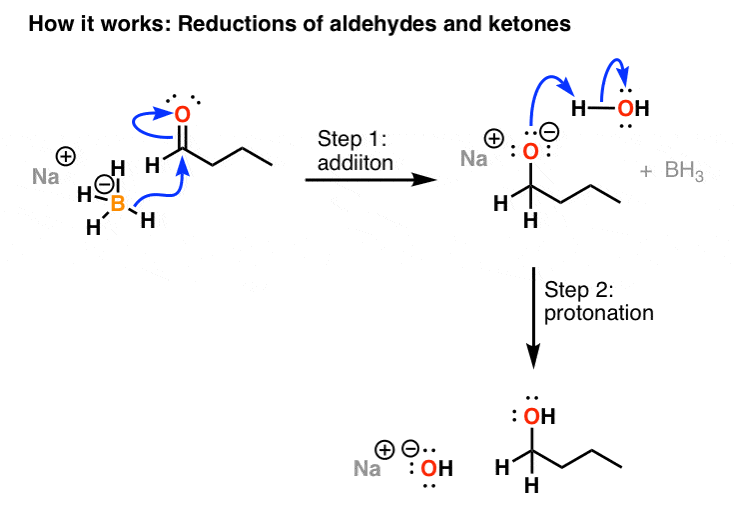
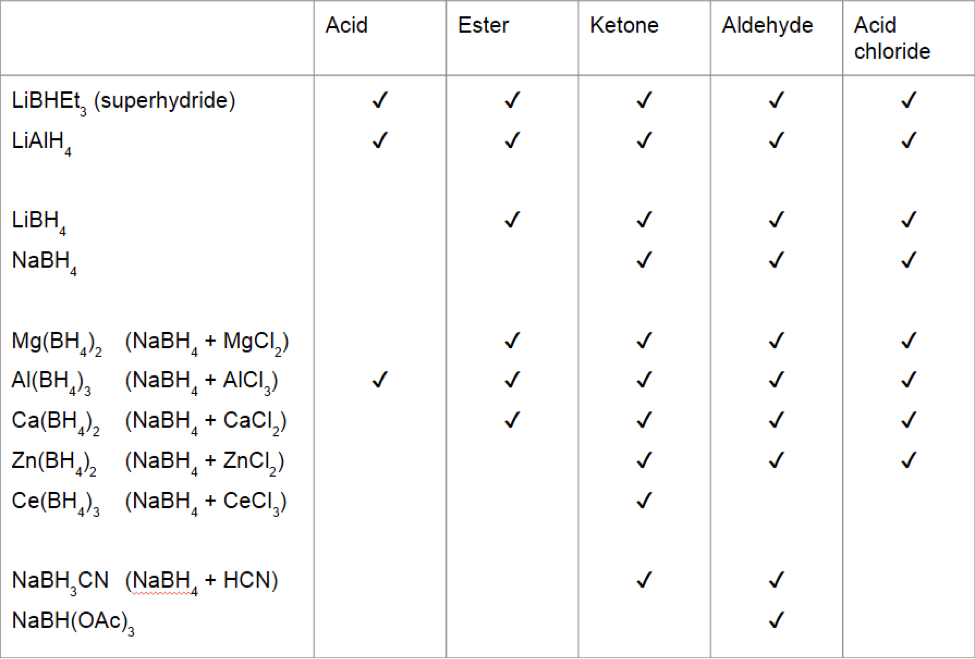
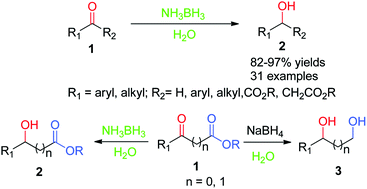
How to reduce carbonyl. Reactions at the carbonyl group of acid derivatives with irreversible nucleophiles just as we saw with aldehydes and ketones, we can reduce a carbonyl group by the addition of a hydride ion. Mammalian cells may induce several mechanisms to protect themselves against the detrimental effects of carbonyl stress. Take the lower lactam carbonyl group to be reduced to a hydroxyl group, trial of lithium aluminum hydride, and sodium borohydride.nothing to react to sodium borohydride is.
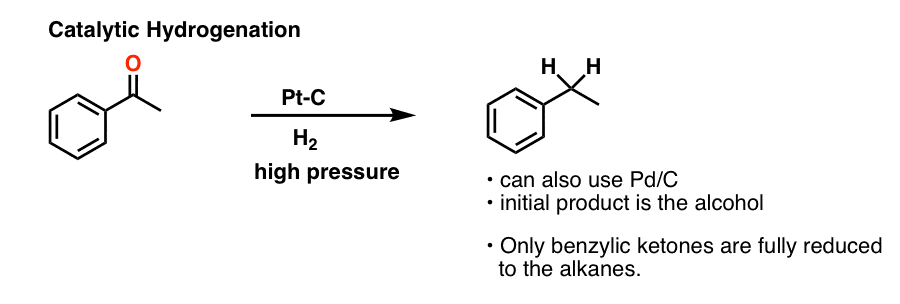
Reduces α, β unsaturated ketones to methylene, and have several feasible methods?trying the sodium borohydride trifluoroacetic acid system can react but not ideally,. The three major pathways to reduce carbonyls to alcohols are catalytic hydrogenation, hydride reduction, and borane reduction. Catalytic hydrogenation is similar to the reduction of an.
Nabh 4 is unreactive enough that the reductions can be done. Sn is not a strong enough reductant to reduce carbonyls (recall reduction potentials from general chemistry). In the case of compounds with ketones (carbonyl groups) and aldehydes.
A compound contains both alpha,beta unsaturated carbonyl group and a normal carbonyl group. Is there any method to reduce the normal. There are many ways humankind can pitch in to help reduce carbon emissions:
The effects of ros are diminished by enzymatic. Various reduction methods for carbonyl compounds converting ketones and aldehydes to alcohol. Of a conjugated carbonyl compound, nabh 4 is not;
Ok, i have this expired 500 g lecture bottle of anhydrous hbr (cost 700 euro).